Entradas
Mostrando las entradas de noviembre, 2014
| Lista de correo. Espere su aprobación. |
| Consultar este grupo |
Chimpanzee Adenovirus Vector #Ebola Vaccine - Preliminary Report.
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
Algae-based oral recombinant vaccines
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
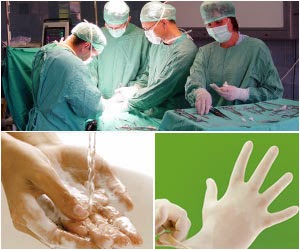
Preventing Health Care–Associated Infections
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
El virus de #Chikungunya
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
How to conduct safe and dignified burial of a #ebola patient
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
COURSE: Guidance for use of Personal Protective Equipment (PPE) During Management of Patients with #Ebola Virus
- Obtener vínculo
- X
- Correo electrónico
- Otras apps
Plant-derived virus-like particles as vaccines
- Obtener vínculo
- X
- Correo electrónico
- Otras apps